Patient Identification Equipment In Europe
-
by STALICLAbased in SWITZERLAND
STALICLA’s precision medicine approach begins with ‘endophenotyping’-identifying the nonbehavioral physical and molecular presentations of NDDs and characterizing subgroups of patients with similar disease signatures. DEPI matches these patient subgroups with an NDD-targeted drug (or drug combination) by integrating and ...
-
Manufactured by IdenPro BVbased in BELGIUM
IdenPro’s i-Band series RFID direct thermal printable wristbands are a crucial part of patient admission system and helps improve its workflow efficiency by connecting patients and the hospitals IT infrastructure. It also helps aid patient ...
-
Manufactured by IdenPro BVbased in BELGIUM
IdenPro’s ProBand series Direct thermal & Thermal transfer printable wristbands with adhesive closure require no assembly, which means printing and securing onto the patients is quick & ...
-
based in UNITED KINGDOM
are visualised as green amplicon peaks. The internal amplification control markers (non-cystic fibrosis) found in CF-EU2v1 are also included in the CF Italia Panel mix. These are visualised as red amplicon peaks and are used to monitor the efficiency of sample amplification and aid patient identification when running the CF Italia Panel alongside the ...
-
based in FRANCE
, for patient input identification, a spare battery, to double the systems autonomy, a backpack for secure transportation to your point of ...
-
Manufactured by La Tecnocarta srlbased in ITALY
Wristbands for medical use are the most reliable solution in terms of patient safety, as they carry personal and clinical information for a proper patient identification, correct treatment administration and accurate sample tracking. Patient data are conveyed by barcodes and QR codes, which can be thermally or laser-printed on ...
-
Manufactured by Cardioline S.P.A.based in ITALY
identification. Light and small: only 100g for maximum patient ...
-
Manufactured by GENFITbased in FRANCE
Nonalcoholic steatohepatitis (NASH) remains largely underdiagnosed, as it carries no obvious symptoms in its early stages, and also because of the lack of widely available non-invasive tests specifically developed to diagnose the disease. The underdiagnosis of NASH represents a major unmet need, and is a barrier for identification of ...
-
Manufactured by Longenesisbased in LATVIA
Enable biomedical data identification with Curator; Biomedical research acceleration platform that provides an opportunity for clinical investigators, patient organisations, registries, biobanks etc. to showcase the scope of data for research without compromising the privacy of the patients and data protection regulations and allowing ...
-
based in AUSTRIA
resources. Electronic registration based on unique drug codes. Personal identification of patient? doctor? pharmacist and medical prescription. Automated Reports. Goals and limits of drug ...
-
Manufactured by MELET SCHLOESING Laboratoiresbased in FRANCE
Used with analysers of the MS9 range, the M-Sampler pierces the cap of the sample tube, reads the patient ID barcodes and transfers the sample to the analyzer for ...
-
Manufactured by Kettering Surgical Appliances Ltdbased in UNITED KINGDOM
Detecto Handrail Stand-on Scales Stainless steel handrail with handgrips and skidproof rubber mat help steady patients. 7⁄10"H LCD display can be read from either side of scale. Functions: patient ID capture, Body Mass Index (BMI), display hold/release, auto zero, motion detection, and auto power off. Operates on either six C batteries or ...
-
Manufactured by Eurospital Diagnostic Division | Eurospital Spabased in ITALY
The first rapid monotest for the determination of anti-transglutaminase antibodies in human ...
-
Manufactured by Selenium Medicalbased in FRANCE
SoBliss® allows new blister packaging vision. Implants can now be packaged in 360° transparent & rigid “double-blister”, that provides a better match in terms of identification, handling, and storage. It also secures transfer procedure (no aseptic faults during surgeries thanks to its “No Touch” ...
-
Manufactured by AVALUN SASbased in FRANCE
LabPad® Evolution is a handheld Point of Care (PoC) multi-measurements testing device, designed to assist healthcare professionals in direct contact with patients, wherever they are. It operates with a range of compatible single-use SmartChip tests, each SmartChip allowing for the performance of a specific biology test. Currently ...
-
Manufactured by Novacorbased in FRANCE
The Vista Access features 24 hours full disclosure ECG on 1, 2 or 3 channels as well as the the traditional Novacor flare for innovative technical function and design ...
-
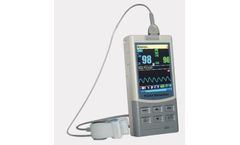
Manufactured by Timesco Healthcare Ltdbased in UNITED KINGDOM
The 300M hand held pulse oximeter can continuously monitor, spot check, replay the data in real time and display Sp02 plethysmograph. It can be widely applied in hospitals and clinics with up to 30 hours of continuous use. It is supplied with 3 x AA batteries and complete with a 3 year guarantee. ...
-
Manufactured by Novacorbased in FRANCE
The Vista Plus features up to 11 days full disclosure ECG on 1, 2 or 3 channels as well as the the traditional Novacor flare for innovative technical function and design. ...
-
Manufactured by HemoCue ABbased in SWEDEN
HemoCue Hb 201 DM System offers the lab accuracy and ease of use that has become a standard in Hb point-of-care testing, plus improved patient safety with features like QC lockout and built-in barcode scanning for patient ...
-
Manufactured by Labitec GmbHbased in GERMANY
Semi-automated 4-channel coagulation analyzer for fast and accurate determination of coagulation, chromogenic and immunturbidimetric assays. Compact in size, easy to operate, this analyzer is the ideal partner for the mid-size coagulation laboratory with slightly increased throughput. The CoaDATA 4004 is the consistent continuing development of the existing turbodensitometric measuring principle. ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you