Risk Of Stroke Equipment
-
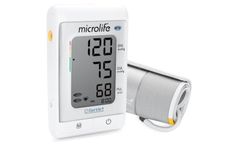
Manufactured by Microlife Corporationbased in SWITZERLAND
The A200 AFIB is equipped with Microlife’s unique AFIB technology, which makes it possible to detect atrial fibrillation while measuring blood pressure at home. Two out of three atrial fibrillation related strokes can be prevented if they were diagnosed early and treated accordingly. Additionally, the PC link function of this device ...
-
Manufactured by Artivion, Incbased in USA
thinner may be reasonable for patients with the mechanical On-X Aortic Valve. In a prospective randomized clinical trial, On-X Aortic Valve patients with a reduced blood thinner dose* had > 60% fewer bleeding events without an increase in risk of stroke. The On-X Aortic Valve, as a mechanical heart valve, has a much lower risk of reoperation ...
-
Manufactured by Arcadia Biosciencesbased in USA
Eating a fiber-rich diet is associated with better gastrointestinal health and a reduced risk of heart attacks, strokes, high cholesterol, obesity, type 2 diabetes, even some cancers. Our high fiber Resistant Starch GoodWheat ingredients contain up to 10 times the dietary fiber of traditional wheat, enough fiber to meet US Food and ...
-
Manufactured by Arkray USA, Inc.based in USA
Highly Accurate1 Blood Glucose Testing in a Feature-focused System. Tight glycemic control can reduce the risk of diabetes-related complications by at least 42%*.2. * 76% reduced risk of eye disease; 50% reduced risk of kidney disease; 60% reduced risk of nerve disease; 57% reduced risk of nonfatal heart attack, ...
-
Manufactured by Randox Laboratories Ltd.based in UNITED KINGDOM
Lipid tests are used to assess an individual’s risk of heart attack or stroke. Lipid tests are the most important of the cardiac risk tests as they provide a clear indication of whether someone is likely to have a coronary event caused by a blockage of the blood vessels or atherosclerosis (narrowing of the arteries caused by ...
-
Manufactured by Omron Healthcare, Incbased in NETHERLANDS
condition which can lead to a higher risk for stroke and embolism. Automatically update your latest results to your smartphone by Bluetooth and have your blood pressure readings at your fingertips through the Omron connect app or you can just ask Alexa to let you know your latest readings without even picking up your phone. Take the hassle out of ...
-
Manufactured by Deltex Medical Limitedbased in UNITED KINGDOM
In collaboration with Lariboisiere Hospital Paris, Deltex Medical has developed a highly innovative method of simultaneously displaying aortic blood flow velocity and aortic blood pressure throughout every heartbeat. TrueVue™ Loops plots aortic blood flow velocity (using oesophageal Doppler ultrasound monitoring) against aortic blood pressure from a standard arterial pressure transducer. It ...
-
Manufactured by Aalto Scientific, Ltd.based in USA
Homocysteine Control is a stable liquid product intended to simulate human patient samples for the purpose of monitoring the precision of laboratory testing procedures for ...
-
Manufactured by Xi’an Ruisaen Biotechnology Co., Ltd.based in CHINA
A structure similar to glucose, vitamin C is a kind of polyol, 2 and 3 in the molecular of two adjacent enol hydroxyl easy to disintegrate and release H +, the reason is the nature of the acid, also known as ascorbic acid. Vitamin C has strong reducibility, is easy to be oxidized to dehydrogenation vitamin C, but the reaction is reversible, and ascorbic acid and dehydrogenation ascorbic acid has ...
-
Manufactured by Viatom Technology Co., Ltd.based in CHINA
A wrist oximeter with long battery life and data capture every two seconds; Screen is bigger and battery life is longer. ...
-
Manufactured by Protembis GmbHbased in GERMANY
ProtEmbo is a catheter-based cerebral protection system to deflect embolic material away from the cerebrovascular circulation during leftsided heart procedures – in particular transcatheter aortic valve replacement ...
-
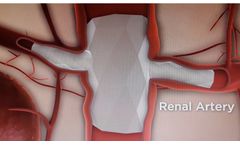
Manufactured by Endospan Ltd.based in ISRAEL
thigh). This delivery method is called an endovascular approach. NEXUS is designed to make a minimally-invasive repair possible for more patients while reducing the risks of ...
-
Manufactured by ACME Portable Machines, Inc.based in TAIWAN
Our BPG 8000 is an automatic blood pressure monitor with AFib Detection. This device accurately detects in real-time irregular heart rhythm from Atrial Fibrillation with our proprietary PPG (photoplethysmogram) technology. Our BPG 8000 can measure both blood pressure and atrial fibrillation at the same time. Within 30 seconds, you will have your results! It's so easy to use that you can use this ...
-
Manufactured by Red Vascular Technology, LLCbased in USA
Red Vascular’s non-modular, branched endoprosthesis represents an ideal solution for aortic aneurysmal disease with branch artery involvement. Its design and delivery through a single femoral artery site are superior to modular, fenestrated grafts currently used to treat complex aneurysms. Fenestration adds several complex steps to deployment of multiple grafts and leads to ...
-
Manufactured by InspireMD Inc.based in ISRAEL
CGuard EPS is designed to prevent peri-procedural and late embolisation by trapping potential emboli against the arterial wall while maintaining perfusion to the external carotid artery. Data from the PARADIGM-Extend study in patients with symptomatic or increased-stroke-risk asymptomatic carotid stenosis demonstrate sustained embolic ...
-
Manufactured by Nyxoah SAbased in BELGIUM
Genio is the disruptive sleep apnea solution for patients who suffer from moderate to severe Obstructive Sleep Apnea or OSA. Genio is the world’s first and unique battery-free, leadless, full body MR conditional and minimally invasive neurostimulator that was designed and developed with users in mind. By stimulating the hypoglossal nerve bilaterally, Genio maintains your upper airway open ...
-
Manufactured by Lumosa Therapeutics Co., Ltd.based in TAIWAN
is tissue plasminogen activator (tPA, also known as IV tPA). tPA works by dissolving the clot but only for those patients presenting within 3 or 4.5 hours of ischemic stroke onset due to increased risk for intracranial hemorrhage in stroke patients with extended time window. Due to its limited applied condition and safety concerns, the actual ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you