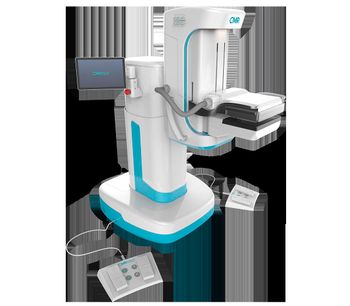
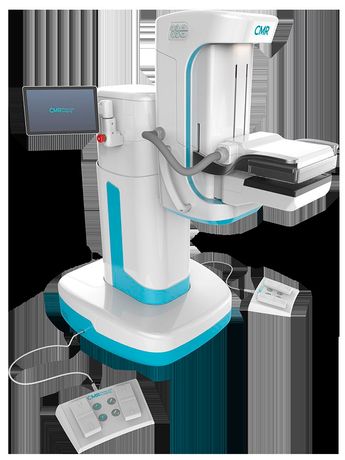
CMR LumaGEM - Molecular Breast Imaging System
The LumaGEM® Molecular Breast Imaging system’s patented mounting mechanism allows the use of light breast immobilization for greater patient comfort during breast cancer imaging exam. The LumaGEM MBI system for breast cancer imaging visually illuminates the enhanced metabolic activity in the breast associated with the occurrence of a tumor growth that is typically not visible on a digital mammogram due to tissue density and/or tumor size.
Details
MBIs are extremely well tolerated by patients, as it’s a patient-friendly and a cost-effective tool for detecting hard to find small lesions in women who have dense breast tissue. When used as a secondary screening modality following digital mammography, the LumaGEM MBI system has increased breast cancer detection rates by almost 400% in early-stage invasive cancers.
- If 1,000 women are screened with traditional digital mammography, 3 women would be found to have cancer.
- If 1,000 women were to be screened with digital mammography and MBI, nearly 12 women would be found to have cancer.
The LumaGEM MBI system for breast cancer imaging is a functional imaging technology that measures the distribution of radionuclides by means of photon detection in order to aid in the evaluation of lesions in the breast tissue. The system features include:
- Clear resolution (high intrinsic spatial resolution, 1.6mm), the best available detector on the market
- Offers clinically high accuracy with Low false negatives (high sensitivity, 91%) and Low false positives (high specificity, 93%)
- Knowing what is negative (Negative Predictive Value, 99.8%)
- Proven/published lower dose capabilities minimizing patient risk (2.6mSv at 8 mCi)
- Patient friendly, comfortable exam with minimal compression
- Alternative for women contraindicated for MRI due to pacemakers or other ferromagnetic implants, poor renal function, claustrophobia, and body size or gadolinium allergies
- Seamlessly integrates into workflow: Looks and acts like a mammography system with easy comparison to standard mammographic views (cranial caudal and mediolateral oblique)
The LumaGEM MBI system received FDA 510(k) clearance in 2011, and is installed in hospitals and breast imaging centers around the country.
What Separates MBI from the Past
What Separates MBI from the Past
Digitizing Molecular Breast Imaging: Earlier versions of this technology were introduced roughly 15 years ago. The LumaGEM represents a significant leap forward in technology and performance with fully digital, dual head camera detectors. With these improvements, LumaGEM customers have been able to identify cancers as small as 5mm in women with dense breast tissue, with greater than 90% sensitivity and specificity.
Awards and Recognition
- Bronze Award Winner in the Radiological and Electromechanical Devices Category of the 17th Annual Medical Design Excellence Awards (MDEA) competition
- Winner: Frost & Sullivan 2014 Best Practices Award – Global Breast Imaging Systems Product Leadership Award
LumaGEM Specifications At a Glance
- Detector Configuration: Dual head
- Detectors: Cadmium Zinc Telluride (CZT)
- Typical Clinical Dose1: 8 mCi (99mTc-Sestamibi)
- Whole-Body Dose2,3: 2.4mSv (at 8 mCi)
- Intrinsic Spatial Resolution: 1.6mm
- Energy Resolution: Full-Width Half-Maximum (FWHM): Sensitivity2: 91% (with mammo)
- Specificity2: 93%
- Collimator: Registered tungsten
- Field-of-View: 16×20 cm2
LumaGEM MBI Workflow
A New Standard of Care in Early Breast Cancer Detection
Even when a mammogram is deemed negative, additional adjunctive tests, such as Molecular Breast Imaging (MBI), for women with dense breast tissue or who are considered high-risk for breast cancer should be considered.
When a patient is determined to have dense breast tissue, or has a suspicious mammogram, MBI offers a secondary screening option for breast cancer imaging that is more sensitive and specific. As a functional imaging modality, MBI provides the ability to have confidence in the diagnosis, and provide patients and physicians alike with peace of mind.
The Ideal MBI Workflow/Patient Algorithm
Molecular Breast Imaging is used as an adjunct/second look modality for dense breast women with lifetime risk of less than 20% after their annual mammograms. The images produced by an MBI exam are the same standard screening views as mammography, cranial caudal (CC) and mediolateral oblique (MLO), and are often read side by side with the patient’s prior mammogram. Users praise the fact that images are read in seconds and describe MBI as the “shortest path to the best care, at the lowest cost.”
With a demonstrated sensitivity and specificity over 90%, and 99.9%, negative predictive value, using MBI for dense breast cancer imaging delivers a strong value proposition to doctors by providing workflow efficiency, improved outcomes, and patient satisfaction.
For the small minority of dense breast women whose risk factors place them at greater than 20% lifetime cancer risk (i.e., BRCA gene mutation, first degree relative with breast cancer, etc.), MRI is generally indicated. For those patients who have a 20% or greater lifetime risk, but are contraindicated and/or cannot tolerate an MRI, MBI also offers a solution for early breast cancer detection and imaging in patients with dense breast tissue.
LumaGEM Procedure Overview
Positioning on the LumaGEM® Molecular Breast Imaging (MBI) system is analogous to mammography for obtaining standard CC (cranial caudal) and MLO (mediolateral oblique) images. Its similarity to mammographic positioning makes LumaGEM an easy modality for any trained mammographer to learn and use, and easy comparison to the original mammography views by the radiologist.
The breast cancer scanning procedure may begin five minutes after the patient has been injected with a small amount of radioactive tracer (Tc-99m Sestamibi). The tracer preferentially accumulates in cancer cells, and is not influenced by breast density.
The patient sits comfortably while the breast is immobilized between two gamma cameras, using just enough pressure to keep it stationary for 5-10 minutes while images are taken. Compression is used only to stabilize the breast adequately, which means that minimum compression is required compared to mammography.
The entire breast cancer screening study takes approximately 40-45 minutes; and images are immediately available for a physician’s interpretation.
Customer reviews
No reviews were found for CMR LumaGEM - Molecular Breast Imaging System. Be the first to review!