

Oncoral - Novel Daily Irinotecan Chemotherapy Agent
Oncoral is a novel daily irinotecan chemotherapy in development. Irinotecan chemotherapy has an established potent anti-tumor effect – even in difficult to treat cancers. Oncoral is a daily irinotecan tablet with the potential to offer better patient outcomes with improved safety following the daily dosing at home compared to intravenous high-dose infusions at the hospital. Following successful Phase 1 results, Phase 2 clinical development for Oncoral is in preparation.
ONCORAL ISA DAILYTABLETCHEMOTHERAPY FORMULATION
Oncoral is a patented tablet formulation of the topoisomerase I inhibitor irinotecan, a chemotherapeutic drug with a well-established role and strong anti-tumor activity in the treatment of various cancer types.
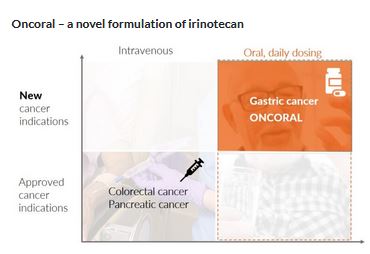
Oncoral is initially being developed for the treatment of gastric cancer in combination with other anti-cancer treatments. Watch presentations about the development plan here:
Irinotecan is a so called antineoplastic agent that after metabolic activation inhibits the enzyme topoisomerase 1, thereby inducing cancer cell death via the prevention of their DNA replication. Irinotecan is converted by carboxylesterases, primarily in the liver, to the active metabolite SN-38 which is 100-1.000 more potent than irinotecan in killing tumor cells.
ONCORAL CAN BE FIRST ORAL VERSION OF IRINOTECAN
Oncoral is a new patented oral tablet formulation of irinotecan in clinical development In Phase 1 clinical studies, the oral formulation has been shown to enable a secure and efficient release and uptake of irinotecan from the gastro-intestinal tract.
Data also shows a high conversion rate of irinotecan to the active metabolite SN-38, which has a high anti-tumor activity.
BENEF1TSTO PATIENTS AND HEALTHCARE SYSTEMS
Treatment with currently available intravenous (IV) irinotecan infusions is often a trade-off between efficacy and tolerabilityforthe patient leading to sub-optimal outcomes for many cancer types, and toxicity related dose-reductions or discontinuations.
In addition, the suppressed immune function stemming from the chemotherapy treatment leads to an increased risk of infections and common gastrointestinal adverse events.
Oncoral is a daily irinotecan tablet with the potential to offer better patient outcomes with improved safety. Daily dosing can potentially improve efficacy driven by a more favorable, dosing-related, pharmacokinetic and pharmacodynamic profile. A continuous, low dose regimen also has the potential to reduce the severity of adverse events, infections, and other complications, including dosing flexibility and the possibility of faster discontinuation.
Furthermore, Oncoral has the potential to be combined with other chemotherapies and cancer drugs to potentially enable an all-oral combination option, which may reduce the burden of treatment and make adherence easier.
In addition, home administration can increase convenience for patients and alleviate healthcare staff and equipment allocated to hospital IV administration.
Following successful Phase 1 results. Phase 2 clinical development for Oncoral is in preparation.
PROMISING RESULTS IN PHASE 1
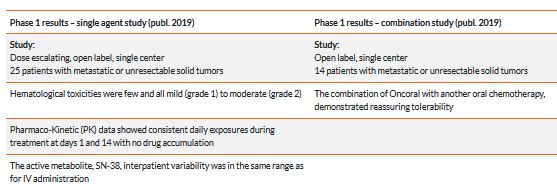
Oncoral has completed a Phase 1 trial at Herlev Hospital, Denmark. Data from the trial shows that Oncoral was well tolerated and confirmed the expected pharmacokinetics of Oncoral given as single agent. The study also established the maximum tolerated dose of Oncoral given as single agent and when administered in combination with another oral chemotherapeutic drug.
A total of 25 patients were enrolled in the first part of the study with Oncoral given as single agent A further 12 patients were enrolled in the second part of the study where Oncoral was given in the combination study.
CLINICAL DEVELOPMENT STRATEGY AND PREPARING FOR PHASE 2
The clinical development strategy for Oncoral is to obtain Phase 2 data providing a clinical proof of concept in gastric cancer.
The plan is to design and conduct a Phase 2 study on Oncoral in combination with other anti-cancer drugs (potentially oral) in patients who are irinotecan-naive and HER2 negative with unresectable or metastatic gastric cancer.
Vou can read more about the Phase 2 plans here
