
OncoStem CanAssist - Prognostic Test
CanAssist Breast is a prognostic test developed by OncoStem that classifies the patients as ‘low-risk’ or ‘high-risk’ based on the patient’s risk of breast cancer recurrence over five years. CanAssist Breast provides information about the risk of recurrence of early-stage, hormone receptor-positive breast cancer patients. This test analyses critical biomarkers in the tumour to assess the risk of recurrence, and helps oncologists to personalize treatment plans.
-
Most popular related searches
Know Your Recurrence Risk
Recurrence risk prediction is an important step towards personalized cancer treatment. In up to 3 out of 4 early-stage hormone receptor-positive breast cancer patients, the benefit from chemotherapy is not outweighed by its risks.
Personalize Your Cancer Treatment
With vital insights gained through the results of CanAssist Breast, it is possible to identify the patient's risk of a cancer recurrence over five years. By understanding the tumour biology, the test calculates the risk of recurrence and classifies the patients as ‘low risk’ and ‘high risk’ using a machine learning statistical algorithm. This enables the doctors to personalize treatment for each individual, resulting in improved quality of life and better clinical outcomes.
Avoid Chemotherapy and it`s Side Effects
CanAssist Breast can precisely identify the need for chemotherapy among early- stage breast cancer patients, by assessing their risk of recurrence. Chemotherapy takes a toll on both physical and mental health of patients undergoing treatment. It is primarily designed to target the fast-growing cancer cells. However, it also destroys healthy cells in its course, causing further debilitating side effects in the patients.
CanAssist Breast determines the patient’s risk of breast cancer recurrence over ?ve years, classifying patients as ‘low-risk’ or ‘high-risk’ for cancer recurrence.The 'low-risk' patients’ can possibly avoid chemotherapy and its side effects and personalize their cancer treatment.
Some of these side effects include:
- Hair loss
- Anaemia
- Nausea
- Diarrhea
- Constipation/Diarrhea
- Fatigue
- Oral Infection
- Kidney Problem
- Weight loss
- Fertility Problems
- Skin & Nail Allergy
Am I Eligible?
The patients must meet the following criteria to take the CanAssist Breast test:
- Patients with early-stage (Stage I or II), invasive breast cancer
- Patients with hormone-receptor-positive (ER+/PR+) and HER2-negative disease
- Patients with lymph node negative (NO) or up to three lymph nodes positive (N1) disease
- Patients who have not gone through neo-adjuvant chemotherapy
If you meet the eligibility criteria, your doctor can prescribe CanAssist Breast post-surgery. Supported with the test results, your doctor can decide on how likely it is that you would benefit from addition of chemotherapy to your treatment plan.
*The test is not applicable for patients diagnosed with DCIS (Ductal Carcinoma In- Situ).
Science and Validation
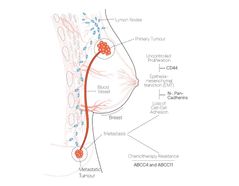
CanAssist Breast integrates IHC (immunohistochemistry) data and clinical parameters to calculate a risk score that classifies patients as ‘low-risk’ or ‘high-risk’ for breast cancer recurrence over five years. By integrating the tumour biology of the disease with time-tested clinico-pathological parameters we assist physicians in developing optimal treatment plans for cancer patients. The CanAssist Breast test result allows breast cancer patients who are classi?ed as ‘low-risk’ to potentially avoid chemotherapy and its side effects.
Overview of Canassist Breast Test Technology
- CanAssist Breast is an immunohistochemistry (IHC)-based novel risk classifier combining five patented biomarkers and three clinico- pathological parameters to calculate risk of cancer recurrence at a site other than breast within five years from diagnosis.
- The 5 biomarkers chosen play critical roles in cancer recurrence.
- The machine learning-based techniques used help assess non-linear interactions between the various signaling pathways that play a role in cancer recurrence.
Analytical Validation
- CanAssist Breast has been rigorously validated based on various international guidelines. Multiple variables that play a role in test precision have been assessed and the test has been found to be repeatable and reproducible
- Stringent QC and validation have been done on all IHC protocols used in the CanAssist Breast test performed in the reference laboratory at Bengaluru.
- CanAssist Breast is CE marked, ISO 13485 certified, and performed in the reference laboratory at Bengaluru
- Our reference laboratory in Bengaluru is accredited by College of American Pathologists (CAP) and NABL, and participates in various External Quality Assessment Schemes (EQAS).
Clinical Validation
- CanAssist Breast has been validated in a retrospective multi-centric clinical study (15+ centres in India, USA & Europe) on close to 2000 patients
- A significant proportion of the validation cohort were aged 50 or younger, making the test suitable for both young pre-menopausal and older post-menopausal patients.
- The validation data has been published in several international peer- reviewed journals like Biomarker Insights and Cancer Medicine:
- Kaplan-Meier survival analysis showed that chemotherapy leads to better results for high-risk patients, but didn’t lead to statistically significant benefit in low-risk patients
- Cox proportional hazards analysis showed that significant prognostic value was provided by CanAssist Breast compared with other traditional clinicopathological parameters
- CanAssist Breast provided significant prognostic value, superior to that provided by traditional clinicopathological parameters and tests such as Ki67 and IHC4.
How It Works?
CanAssist Breast Test examines the activity of proteins present in the cancerous tumour to predict the likelihood of the cancer returning post- surgery. It also helps determine whether chemotherapy will benefit the patient or whether just hormone therapy by itself is sufficient. So patients should not hesitate to ask their doctors how CanAssist Breast Test results may impact their treatment plan. CanAssist Breast provides additional information about the tumor, based on which the cancer treatment can be customized.
Process and Timelines
Once your doctor prescribes CanAssist Breast, the hospital laboratory will send your tumour sample (FFPE blocks) to OncoStem’s laboratory. Our lab performs the test and shares the report with your doctor within ten business days.
- Surgery completed
- Doctor prescribes CanAssist Breast
- Payment for the test completed
- Hospital or patient sends tumour block to OncoStem
- CanAssist Breast performed
- Report generated within 10 business days and sent to doctor
- Doctor will plan further treatment based on the report and other patient-specific parameters
Understand Your Risk Score
If you have been recently diagnosed with early-stage invasive breast cancer, there is a possibility that you may not require chemotherapy. CanAssist Breast test provides additional information about the tumour. Along with the existing pathology report, this test result lets you know the likelihood of cancer returning at a distant site after surgery and whether you are likely to benefit from chemotherapy or not.
Knowing the patient's recurrence risk allows the doctors to devise treatment plans that are in-tune with the prognosis, thereby, maintaining an optimal balance between the benefits and side-effects of chemotherapy.
The CanAssist Breast risk score ranges from 0 -100. A risk score cut off of 15.5 is used to classify patients as low-risk (score 15.5 or less) for distant recurrence. The maximum probability of distant recurrence for low-risk patients for five years is 9% which means that you have less than a 1 in 10 chances of distant cancer recurrence.
Customer reviews
No reviews were found for OncoStem CanAssist - Prognostic Test. Be the first to review!


