Monofix - Model PDO, PGCL - Absorbable Barbed Sutures
From Medical Devices - Suture - MIS Sutures
Absorbable barbed sutures with a needle used for closing general soft tissues.
Product Details
Product Name
- Monofix® PDO (polydioxanone suture)
- Monofix® PGCL (poliglecaprone suture)
Raw Material
- Monofix® PDO: polydioxanone [poly(1,4-dioxane-2-one)]
- Monofix® PGCL: poliglecaprone [poly(glycolide-co-ε-caprolactone)]
Purpose of Usage
- Absorbable barbed sutures with a needle and stopper are used for closing general soft tissue.
Certification
- Monofix® PDO: CE (Class?)
- Monofix® PGCL: FDA (Class?)
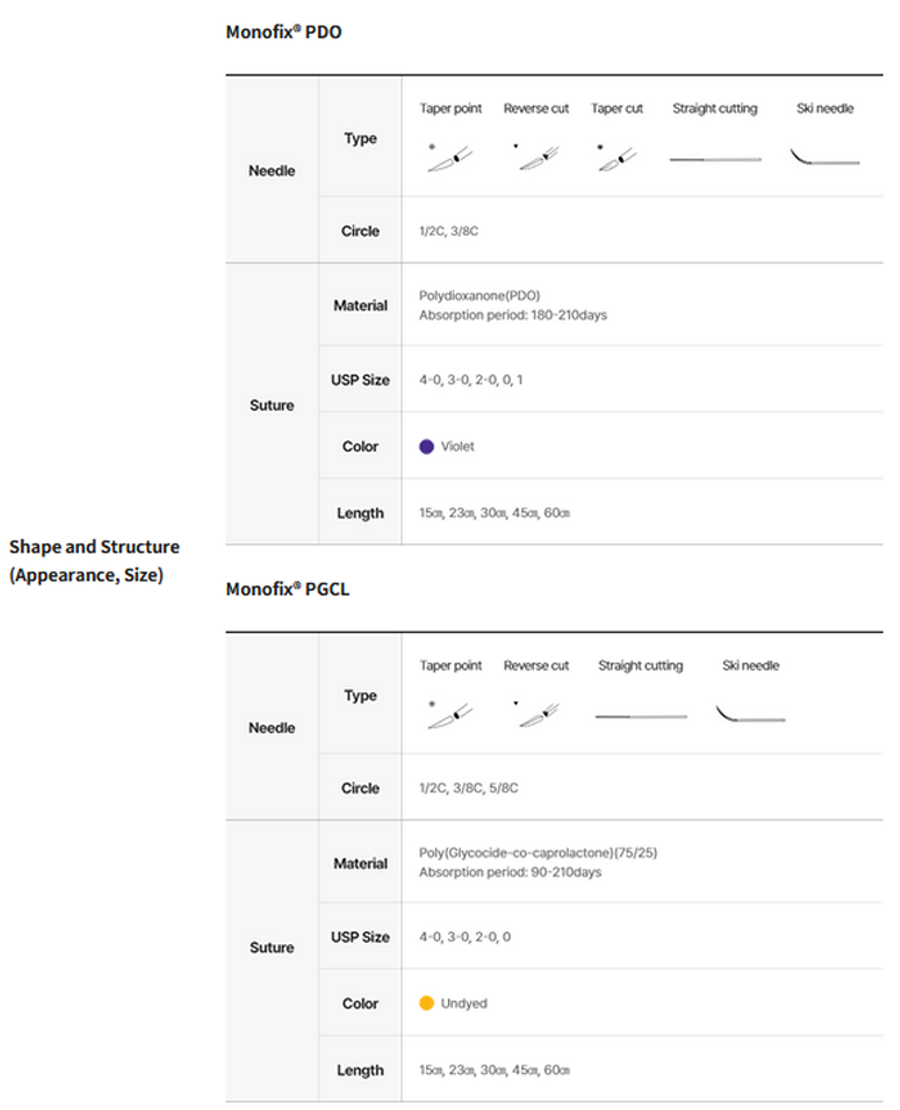
Shape and Structure
Shape and Structure
(Principle of Action)
Monofix® PDO: This product is an absorbable monofilament suture made of polydioxanone [poly(1,4-dioxane-2-one)], which is a biodegradable material, and is absorbed into the body through hydrolysis. The animal (rat) implantation test showed that the tensile strength retention rate was approximately 85-89% at two weeks, approximately 70-75% at four weeks, and approximately 40-47% at six weeks after the implantation. The suture was absorbed 180 to 210 days after the implantation.
Monofix® PGCL: This product is an absorbable monofilament suture made of poly(glycolide-co-ε-caprolactone) (PGA-PCL), which is a biodegradable material. It is absorbed into the body through hydrolysis, accompanied by reduced tensile strength and weight. The animal (rat) implantation test showed that the tensile strength retention rate was approximately 68-73% at one week and approximately 33-39% at two weeks after the implantation. The suture was absorbed 90 to 120 days after the implantation.
Package Unit
- 12EA/BOX
Storage Method
- Store away from high temperature, high humidity and direct sunlight.
- Store in a well-ventilated place at room temperature (1~30?) without opening the package and maintain an aseptic state.
Customer reviews
No reviews were found for Monofix - Model PDO, PGCL - Absorbable Barbed Sutures. Be the first to review!