Zygel - Model ZYN002 - Cannabidiol Gel
Zygel is the first and only pharmaceutically-produced cannabidiol, a non-euphoric cannabinoid, formulated as a patent-protected permeation-enhanced gel for transdermal delivery through the skin and into the circulatory system. Zygel is being developed for patients suffering from FXS, ASD, and 22q. Zygel is an experimental treatment. This means that it is not approved by government regulatory bodies, including the United States Food and Drug Administration (FDA) and other agencies and must be tested to see if it is an effective and safe treatment.
Addressing Limitations of Current Treatments
Challenge: Oral administration
Oral administration can lead to limitations in safety and efficacy including gastrointestinal side effects like nausea, vomiting and diarrhea, low bioavailability, inconsistent plasma levels and significant first-pass liver metabolism. First-pass liver metabolism refers to the process by which the liver breaks down drugs ingested directly or indirectly through the gastrointestinal system, such as through oral or oral-mucosal delivery methods, allowing only a small amount of drug to be absorbed into the circulatory system.
Potential Benefit of Zygel:
Zygel is a unique permeation-enhanced CBD transdermal gel. Transdermal therapeutics are applied locally and absorbed through the skin directly into the systemic circulation, resulting in a lower incidence of gastrointestinal side effects, and avoidance of first-pass liver metabolism, which potentially enables lower dosage levels of active pharmaceutical ingredients and rapid, reliable absorption with increased bioavailability. Other potential benefits of transdermal delivery compared with oral administration may include fewer drug-drug interactions.
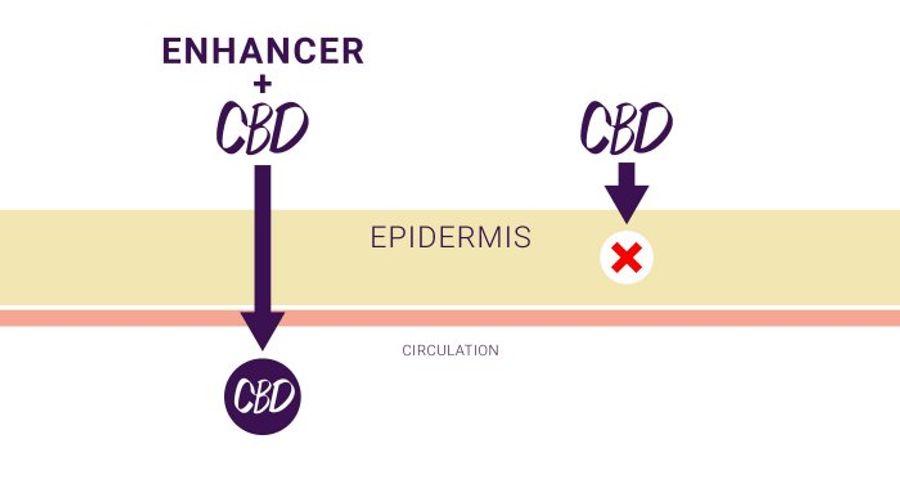
Challenge: Topical Administration
CBD and other cannabinoids are large, complex molecules that do not penetrate the skin efficiently without the use of technologies that improve their penetration. As such, topical formulations of CBD or oils derived from Cannabis may require very high doses to reach therapeutic blood levels, or may not cross the skin and into the bloodstream at all.
Potential Benefit of Zygel:
Zygel is a proprietary pharmaceutically-manufactured CBD gel formulated with permeation enhancers, which improve the ability of CBD to cross through the skin and into the bloodstream when administered directly to the skin (transdermal delivery).
Challenge: CBD degradation to THC in the stomach
An in vitro (performed in a lab in a test tube, culture dish, or elsewhere outside of a living organism) study, titled “Identification of Psychoactive Degradants of Cannabidiol in Simulated Gastric and Physiological Fluid,” which was conducted by Zynerba and published in April 2016, demonstrated that CBD is degraded to THC in an acidic environment such as the stomach. THC is the principal psychoactive cannabinoid. As such, we believe that degradation of CBD into THC may lead to increased negative psychoactive effects.
Potential Benefit of Zygel:
Transdermal delivery of Zygel avoids the gastrointestinal tract and potential degradation to THC in stomach acid, which should minimize the risk of negative psychoactive effects.
Challenge: Botanically derived cannabinoids
Botanical cannabinoids may create significant challenges for drug manufacturers because of the natural resources and security measures required to grow and harvest Cannabis.
Potential Benefit of Zygel:
Zygel is pharmaceutically manufactured in a cGMP (Current Good Manufacturing Practice) facility that will comply with rigorous efficacy and safety standards established by global regulatory agencies and is not extracted from the Cannabis plant. We believe that producing CBD this way ensures that we can provide consistent potency and efficiently scale manufacturing to meet the needs of the markets that we are targeting, without the cost and logistical requirements of growing and harvesting Cannabis.
Customer reviews
No reviews were found for Zygel - Model ZYN002 - Cannabidiol Gel. Be the first to review!