

- Home
- Companies
- BrightInsight, Inc
- Software
- BrightInsight - Regulated Digital ...
BrightInsight - Regulated Digital Health Platform Software
The leading global regulated Digital Health platform for biopharma and medtech.
Built under a Quality Management System to support regulated products, our medical-grade BrightInsight™ Platform uses software and services to capture, transmit and analyze data from connected medical devices and surgical equipment, combination products, apps, Digital Therapeutics and Software as a Medical Device- all in compliance with security, privacy and regulatory requirements.
BrightInsight offers connectivity and management for regulated apps, algorithms, surgical equipment and medical devices, combination products or Digital Therapeutics.
BrightInsight can aggregate data from multiple sources and can integrate with EHRs and other IT systems.
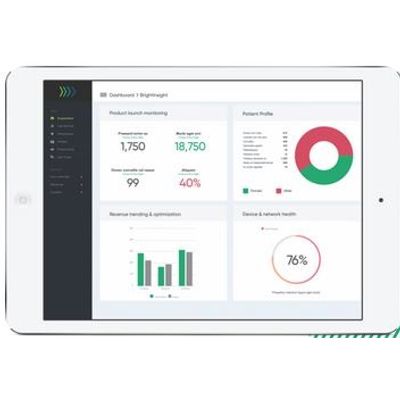
Clinical, operations and patient engagement dashboards provide valuable insights in real-time.
- BrightInsight supports CE-Marked and FDA-regulated class I, II and III medical devices, combination products and Software as a Medical Device (SaMD)
- Our cutting-edge Quality Management System is ISO 13485:2016 certified
- We provide compliant change control for the continuously evolving cloud infrastructure
- BrightInsight’s Device Master File has been accepted by the FDA and can simply be referenced in your regulatory submission
- A device-agnostic solution, BrightInsight can easily support multiple products
- The microservices architecture provides advanced security, scalability and customization options
- Built with clinical workflow integration in mind, BrightInsight integrates with Electronic Health Records (EHRs) and other Health IT systems
- Ongoing operational and administrative security safeguards including response and forensics analysis, regular vulnerability assessment and risk analysis, regular internal and third-party penetration testing, security updates and patching, regular testing for disaster recovery, and regular third-party audits
- Defense-in-depth architecture with multiple dimensions of technical controls
- Privacy compliance support, including audit logs, data retention, incident response and breach notifications, Data Protection Impact Assessment, and patient consent management.
- BrightInsight generates real-time insights from real-world drug and device data
- Our Engagement Dashboard provides analytics to enable proactive patient communication and data-driven app optimization
- Our Adherence Dashboard provides drug and device utilization data to support real-world evidence of drug adherence
- Our Operations Dashboard provides insights to track commercial KPIs for product launches, revenue trending, patient demographic information and more
