Clinical Data Software In USA
-
based in USA
Veeva CDB is a clinical data platform that aggregates and harmonizes all data sources so you get clean, well-organized data that is ready-to-use ...
-
based in USA
Ofni Clinical is a clinical data management tool that rapidly creates and implements secure, regulatory compliant clinical trial databases. Ofni Clinical includes comprehensive search and report features, has strong data validation and edit check options, contains flexible customization tools, ...
-
based in USA
Veeva Vault Coder provides an intuitive interface and innovative coding tools that yield fast and accurate coding for any clinical trial. Vault Coder supports coding clinical terms for data on trial medications, adverse events, prior surgeries, medical histories, and more based on MedDRA or WHODrug ...
-
based in USA
Vault EDC for clinical trials provides a fast and intuitive interface for capturing and reviewing data from sites. Eliminate constraints and run the trial you want with a modern EDC ...
-
based in USA
Simplify your data life cycle to enable alignment and unlock efficiency across your clinical ...
-
based in USA
Organizations with on-going needs for biospecimens and clinical data may subscribe to CDNR platform to plan studies, identify patient cohorts for biospecimen collections and real-world data studies, and to manage biospecimen and associated clinical data ...
-
based in ITALY
WebBioBank is a platform specifically developed to collect and analyze neuronal signals integrated with clinical data recorded from patients undergoing neuromodulation treatments, in particular deep brain stimulation (DBS). WebBioBank includes neuro-biosignals and clinical data collected. WebBioBank now integrates neuronal ...
-
by Ovationbased in USA
This Ovation IBD Genomic and Clinical Linked Data sample dataset is a packaged, real-world observational cohort of 10 Inflammatory Bowel Disease (IBD) subjects. It's available at no cost for scientists interested in evaluating Ovation’s Whole Genome Sequencing and RNAseq data with up to 30x coverage. The data is linked with ...
-
Manufactured by Syncro Medicalbased in USA
Cutting edge medical instrument manufacturers are uncovering the hidden value of the data collected and generated by their systems. With Syncro Medical’s help, they’re using this data to gain new insights, both Clinical and Operational. Clinical healthcare analytics focus primarily on the patient, for example patient ...
-
based in USA
OrthoGrid Hip AI is a non-invasive navigation App that enhances existing fluoroscopic data and provides orthopaedic surgeons with valuable, real-time image analysis tools for intra-operative decision-making and execution of both posterior & direct anterior approach ...
-
based in USA
The Etiometry Platform is an end-to-end data management software solution for the collection, analysis, visualization, and archiving of ICU clinical data. It is designed to facilitate the use of all available data to support the anticipation and management of the dynamic condition of patients requiring intensive ...
-
based in USA
Improving The Human Condition Through Data Innovations In Genomics; As an integrated platform, Triton™ brings together clinical data sources in a fully-scalable and customized data warehouse. Leveraging this powerful technology stack, our clients realize actionable and comprehensive results to improve ...
-
based in USA
Etiometry’s T3 Software is designed to fully integrate all available patient data into the clinical decision-making process. T3 drives the fundamental tasks of comprehensive data collection, data presentation, and archiving of patient data. A flexible clinical data ...
-
Manufactured by Carrot Medicalbased in USA
Record Medical Cases for Review rchive Clinical Data ive Stream for Healthcare Educators. Live-stream the data from any medical device to any screen with ...
-
by TriNetX, LLCbased in USA
of millions of patient records from a global EHR network, insurance claims, lab results, and other clinical data in addition to normalized data sets from FDA Adverse Event Reporting System (FAERS), VigiBase, EVDAS, and internal ICSR databases. Enable more efficient detection, refinement, action, and assessment to provide faster answers to drug ...
-
based in USA
Digital Metadata Governance & Clinical Regulatory Compliance: Management and governance of CDISC and corporate standards driven metadata, data models and study level models from protocol to submission with workflows and controls. Solution for clinical trial data submissions in CDISC SDTM to comply with FDA and PMDA mandates. ...
-
based in USA
NLP-based AI for automated mining of clinical documents and reports.Manually extracting clinical data from unstructured text is inefficient. And yet, clinical staff often dedicate up to 30% of their working day repeating just this kind of exercise, and report low levels of satisfaction accordingly. ...
-
based in USA
The right clinical data, at the right time, on any device providing the power of insights beyond just numbers. Streamline data collection and management in real time to ensure transparency, data integrity and quality of your data integrated with your CTMS ...
-
based in USA
computational pathology with exclusive clinical data cohorts, Paige rapidly uncovers and validates phenotypic patterns that are not identifiable using existing methodologies and deploys the AI globally. ...
-
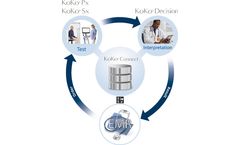
Manufactured by KoKo PFTbased in USA
KoKo Decision presents all relevant clinical and diagnostic respiratory data together in one place for enhanced interpretations with high confidence of accuracy. And to maximize efficiency, the powerful KoKo Connect interface engine makes integrating with your EMR simple and ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you