ISO 13485 Software Available In Africa
-
by Vintarabased in USA
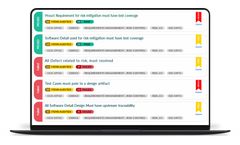
Vintara’s Enterprise Portal helps leaders deploy world class best practices for the manufacture and sale of medical devices. Our configurable workflow applications will drive performance and world class regulatory compliance including: ISO 13485, 21 CFR Part 11 compliance, 21 CFR Part 820 compliance, CAPA, Device Master Records, ...
-
Manufactured by Softeq Development Corp.based in USA
Turn to Softeq to design IoT healthcare devices along with their supporting software: full-fledged operating systems, drivers, cloud-based data analytics tools, medical device interfaces, and mobile apps. Our team of experts will help you navigate the stringent healthcare security standards, including FDA, HITRUST, HIPAA, HL7, DICOM, and ISO ...
-
based in USA
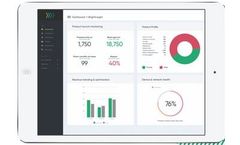
AWARE™ Critical Care provides predictive analytics in healthcare services. AWARE reduces information overload by filtering for relevant patient data and providing best care practices for improved patient outcomes. The AWARE™ software platform has achieved FDA Class II clearance, CE Marking, and ISO 13485:2016 ...
-
Manufactured by Syncro Medicalbased in USA
Mobile is the new frontier for medical instrument innovation. The FDA describes current applications as “just the tip of the iceberg in a coming revolution in mobile medical applications.” We agree, and we stand ready to help your company be a leader in this ...
-
based in GERMANY
Use worldwide knowledge to reliably determine the risk of genetic disease. MH Guide/Mendel is optimized for the evaluation of germline variants associated with hereditary diseases. The software application is a module of MH Guide and supports human geneticists in the evaluation of germline variants. Just like MH Guide, MH Guide/Mendel analyzes gene variants in comparison with data from Dataome, ...
-
Manufactured by Renishaw plcbased in UNITED KINGDOM
Simplifying and streamlining the production of customised CMF/OMF implants and guides. No two patients are the same, so why should their implants be the same? Customised implants allow for personalised healthcare, helping to ensure each patient gets the very best ...
-
based in ISRAEL
Adhere to strict MDR/FDA regulations and ISO quality standards by eliminating paper-based QMS ...
-
Manufactured by Syncro Medicalbased in USA
Whether your product is a wearable, implantable, or a classic bedside monitoring system, Syncro’s long-standing experience developing monitors is a powerful benefit to you and to your ...
-
based in USA
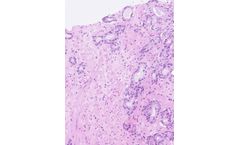
HALO Prostate AI, from Indica Labs, is a CE-IVD certified deep learning based screening tool designed to assist pathologists in the identification and grading of prostate cancer in core needle biopsies. The algorithm automatically analyzes all appropriate case slides and notifies pathologists of cases with suspected findings directly in their native ...
-
based in USA
Document change control software is essential for ensuring compliance and managing change in regulated industries such as life sciences and manufacturing. By utilizing this software, companies can effectively address the complexity of change control and streamline their ...
-
Manufactured by Nextern Incbased in USA
We are your sole partner to provide seamless connected medical device development solutions. Leveraging our experience and expertise in medical device design, cloud architecture, and mobile engagement ...
-
based in USA
The leading global regulated Digital Health platform for biopharma and ...
-
Manufactured by Visarisbased in SERBIA
Visaris LabIS is a latest generation laboratory management and quality control information system for completely paperless automated laboratory and quality system workflow support. It is intended for the use in diagnostic and process laboratories in a wide range of applications including industrial, chemical, food and beverage, veterinary and medical ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you