Medical Device Software
-
based in GERMANY
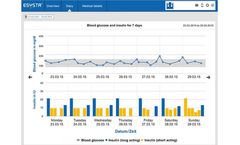
The ESYSTA Portal seamlessly displays the blood glucose values, carbohydrate units and insulin doses sent to it. Special algorithms analyse each patient’s data and evaluate it continuously. A quick summary of critical values or incorrect doses, coupled with rapid communication between the doctor and the patient, brings a new dimension of quality to the treatment of ...
-
by Epihunter NVbased in BELGIUM
Epihunter Pro makes the automatically built seizure diary available through a webportal for the physician. The epihunter home-care solution is based on a wearable EEG headband and a smartphone with a detection algorithm. When a seizure is detected, the people around are notified immediately. Next to this, an entry is automatically logged in the seizure diary. Also, a video can be automatically ...
-
based in USA
We are a regulated Software as a Medical Device (SaMD)-focused biotechnology company that develops our applications as medical treatments in accordance with all relevant FDA, ISO and IEC ...
-
based in GERMANY
Rapidly generate real-world evidence of effectiveness with our modular infrastructure for continuous collection of real-world data. Our RWE generation infrastructure bridges the gap between clinical trials and the real world. Get the right data at the right time while creating tangible value for hospitals and ...
-
based in USA
The medical device reporting (MDR) requirements of 21 CFR 803 apply to manufacturers, importers, distributers, and device user facilities in the United States. Such facilities are required to report adverse device-related events to the Food and Drug Administration (FDA) or the device manufacturer, and ...
-
based in USA
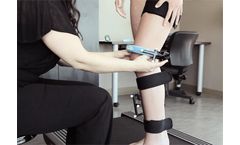
The challenge: Emovi develops and manufactures the KneeKG system, a medical device that assesses knee motion for patients with impaired movement due to an orthopedic cause. The company wanted to modernize some of its software and develop a cloud-based clinical portal without compromising security or reliability. Our solution: We modernized a ...
-
based in CANADA
100% software solution that integrates vital signs information from medical devices with the patient record. Enovacom Patient Connect is a unique connectivity solution that extracts vital signs and other patient information from biomedical devices and integrates this directly with the patient record. This 100% software-based solution ...
-
based in ISRAEL
Our medical requirements management tool is perfect for manufacturers of medical devices and will provide you with a number of essential quality-related ...
-
Manufactured by AB Sciex Pte. Ltdbased in USA
Analyst MD Software: Rely on the Proven Performance for High-Quality Quantitation on your SCIEX IVD LC-MS/MS. Modern clinical laboratories are increasingly opting for mass spectrometry as their preferred sample analysis method. To aid the easy application of mass spectrometry, these labs need software that is intuitive and accessible to both novice and expert ...
-
based in USA
The Sparta risk management solution enables companies to link risk management processes to other quality management processes. This ensures that nonconformities, regardless of source, are evaluated using consistent criteria and standards such as ISO 14971. Additionally, it can trigger updates to review the product risk register to have a truly closed-loop process. The solution is fully ...
-
Manufactured by AMD Global Telemedicine, Inc.based in USA
The Digital Video Colposcope system is a powerful high definition camera and imaging module used for clinical examinations of the cervix, vulva and vagina. The SONY high definition CCD imaging module and super bright LED light source, provide high quality images for clinicians to see pathology changes. This video colposope is different from traditional optical colposcopes because instead of ...
-
Manufactured by AMD Global Telemedicine, Inc.based in USA
The Intraoral Dental Camera is a high powered digital camera used in the evaluation of patients’ oral health during healthcare assessments, and part of the AMD Global Telemedicine family of telehealth products and ...
-
Manufactured by AMD Global Telemedicine, Inc.based in USA
The Digital Wireless ECG is a PC based 12-Lead EKG system that wirelessly streams ECG data from the patient to your personal computer. The process is simple: perform the patient hookup, input patient data into the software, and then start the analysis. Our system uses the Glasgow 12-Lead algorithm to output fully interpretive ECG statements. Reports include Global Measurements, Individual Lead ...
-
Manufactured by AMD Global Telemedicine, Inc.based in USA
The Multipurpose Camera and Scope simplifies clinical telemedicine patient exams and workflow processes. This multi-function camera and medical scope system integrates directly with our AGNES Connect telemedicine platform, delivering a seamlessly integration to capture and stream your medical images to the remote provider. This single ...
-
Manufactured by Covartimbased in BELGIUM
A standalone software, such as a mobile or web application, can fall within the definition of a (IVD) medical device. Therefore, the placing on the market is well regulated and specific actions need to be ...
-
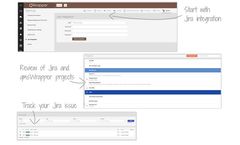
by qmsWrapperbased in CANADA
The combination of Jira with qmsWrapper introduces a system in which MedDev companies are able to track, manage, and organize all CE Technical Files and FDA Design History Files, while enabling software development of medical ...
-
by Metecon GmbHbased in GERMANY
As an alternative to stand-alone software, there are medical devices in which the software is an integral part of the device, so-called embedded ...
-
Manufactured by Shoulder Innovationsbased in USA
Genesis is an early stage investor in medical device technology. We are differentiated through our deep level of engagement in delivering value through all stages of the commercialization process. This is accomplished by leveraging the time, talent, and financial resources of the Genesis Innovation Group Portfolio. ...
-
based in USA
Small Medical Device Companies Can Now Manage and Automate their Design Control Process to Ensure Compliance with 21 CFR Part 820 with the Help of MasterControl's Design Control Software Systems at a Low Monthly Cost. Each manufactured medical device must be supported by appropriate documentation which demonstrates that its ...
-
by Bayoonet AGbased in GERMANY
Digital health applications are CE-marked medical devices that can be prescribed to patients with statutory health insurance. They are used in the diagnosis and treatment of ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you