Medical Device Quality Software In USA
-
Manufactured by Esaote S.p.Abased in ITALY
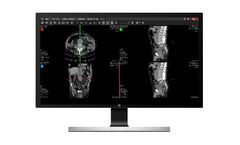
SUITESTENSA UniViewer is the web module dedicated to the distribution of images and reports inside and/or outside the company infrastructure. With a web-based zero-footprint architecture, it is designed to be used as a universal viewer of diagnostic-quality medical images, supporting any device, without any loss of quality. ...
-
based in USA
are supplemental to the quality system requirements of 21 CFR 820 in Quality Suite MEDICAL DEVICES Good Manufacturing Practices for Medical Devices ...
-
based in USA
OOS Software to Automate Out of Specification (OOS) Processes to Comply with FDA CGMPs and 21 CFR Part 211 and Part 820 Regulations. In the FDA environment, specifications are essential in maintaining quality. CGMP regulations for finished pharmaceuticals (21 CFR Parts 210-211) and medical devices (21 CFR Part 820) require strict ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you