

QyScore - Groundbreaking Neuroimaging Analysis Platform Software
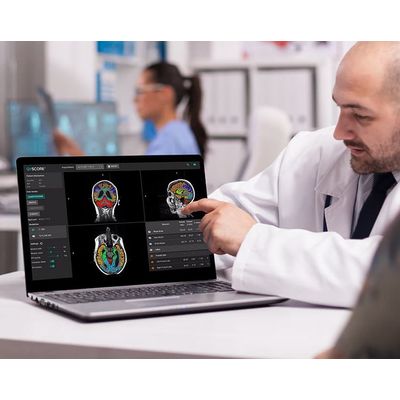
QyScore is a groundbreaking neuroimaging analysis platform for both clinical routine and clinical trial settings. One scan at a time, QyScore has the potential to revolutionize the treatment of CNS disease and create the potential for earlier precision on the frontlines of CNS disease. QyScore is intended for automatic labeling, visualization and volumetric quantification of segmentable brain structures and lesions from a set of MR images. Volumetric data may be compared to reference percentile data. QyScore is not intended for use in clinical scenarios that require evaluation of the number of the white matter hyperintensities.
Designed to seamlessly integrate with existing workflows, QyScore® provides insightful precision for analysis by offering high-quality data that cannot be found through manual examination of a brain scan alone.
Currently deployed in US & EU clinics (expanding soon into new global markets) and used in multi-site clinical trials and global research collaborations, QyScore® is FDA-Cleared, CE-Marked and Qynapse is ISO-Certified.
Powered by robust technology, our platform produces a wide range of automated measures of neuroinflammation and neurodegeneration, such as whole brain, white matter, grey matter by lobe, cerebellum, white matter abnormalities and brain substructures. To facilitate review by physicians, an automated patient report is rapidly generated after each QyScore® analysis and results are viewable in our proprietary and integrated 3D visualization platform.
QyScore® decreases image reading variability and segmentation errors by 89% and has demonstrated better performance compared to certain other state-of-the-art automated methods on the market, promising precision and accuracy.
Peace of mind for routine and research by providing results you can trust.
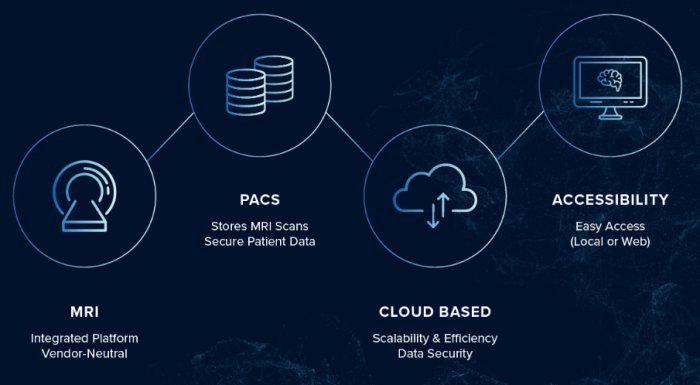
QyScore® gives clinicians the potential to offer more accurate diagnosis and monitoring for patients, and the potential to bring enhanced trial design, improved patient selection, and endpoint protection to clinical trials. Our platform is cloud-based, easy to use and scales to fit your patient load or trial size.
- Support timely diagnosis
- Support disease & therapy monitoring
- Support tailored treatment plans
- Facilitate therapeutic switching
- Potential to enhance trial design, improve patient selection and protect imaging endpoints
- Automates image pre-processing, quality control and image analysis
- FDA-Cleared, CE-Marked platform has the potential to support the contiuum of care from drug development to post-market and long-term monitoring of therapeutic effectiveness and safety.
- Alzheimers and other dementias
- Multiple sclerosis
- Parkinson’s and movement disorders
These disease areas commonly use imaging and automated measurement techniques to help better characterize patients
- Ischemic Stroke
- Huntington’s Disease
- Epilepsy
- Traumatic Brain Injury
- Schizophrenia
- Depression
These disease areas are ones in which we are accumulating data for future use in clinical and research settings.
