Post-Operative Care News
-
Tris Pharma Presents Data Reinforcing Limited Abuse Potential of Investigational Pain Therapy Cebranopadol at the 2023 PAINWeek Conference
Positive results confirm findings from prior studies evaluating the abuse potential of cebranopadol, further highlighting its potential as a safe and potent alternative to traditional opioids Registrational phase 3 clinical trials of cebranopadol in moderate to severe acute pain are on track to initiate in Q4 2023 – Business Wire – Tris Pharma, Inc. (Tris), a commercial-stage ...
-
Creative Diagnostics Launches New Reagent Solutions for Herpesvirus Research
Creative Diagnostics, a leading manufacturer and supplier of antibodies, antigens and assay kits, has announced the launch of new reagent solutions for herpesvirus research, including a range of Human Herpesviruses Antibodies designed to help researchers better understand the structure of Herpesvirus entry and develop new treatments and prevention strategies. The Herpesviridae are a large family ...
-
Tris Pharma Expands Leadership Team as Company Broadens Commercial Portfolio and Progresses Clinical Pipeline
Thomas Englese, a 20+ year industry veteran with a track record of success at leading biopharmaceutical companies, joins as chief commercial officer Marc Lesnick, Ph.D., appointed as chief development officer, bringing more than 20 years of experience in multiple therapeutic areas Tris Pharma, Inc. (Tris), a commercial-stage biopharmaceutical company focused on ADHD, pain and neurological ...
-
Press release: High quality, cost effective surgical retractor is welcomed by leading US hospital group
JUNE Medical is delighted to announce a new agreement with a major US hospital group to supply the award-winning Galaxy II® self-retaining surgical retractors to over 1,600 hospitals in the US, commencing April 15, 2023. This game-changing device radically improves the safety and ease of surgical retraction for both patients and medical staff; its ergonomic design transforms the way in which ...
-
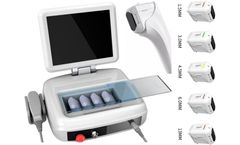
Post-treatment care after HIFU ultrasound cosmetic treatment
HIFU ultrasound machine is a non-surgical SMAS facelift procedure that uses non-invasive high energy focused ultrasound (HIFU) to stimulate the skin's natural regenerative and repair response, tightening the subcutaneous fascia layer and allowing the skin to regenerate collagen on its own, remodelling the elastic fibres and forming a strong elastic support network, thus achieving a strong ...
-
Targeting Alternative Opioid Receptor Reduces Drug Side Effects
A new study published in Proceedings of the National Academy of Sciences (PNAS) indicates, “Delta opioid receptors have a built-in mechanism for pain relief and can be precisely targeted with drug-delivering nanoparticles, making them a promising target for treating chronic inammatory pain with fewer side eects.” Researchers used cells from humans and mice with inammatory bowel ...
-
Prolonged Analgesia From Bupisome And Bupigel Formulations: From Design And Fabrication To Improved Stability
Based on data from early animal studies, Probudur has indicated post-operative control for up to 96 hours, which is 24 hours longer than the leading product on the market. If we are able to demonstrate a successful Phase III clinical trial, we believe Probudur may represent the first long acting local anesthetic with an opioid sparing label. ...
-
Eagle Pharmaceuticals Provides Business Update and Guidance for 2023
Eagle Pharmaceuticals, Inc. (Nasdaq: EGRX) (“Eagle” or the “Company”) today provided a business update and guidance for 2023. Highlights: During the 12 months ended September 30, 2022, Eagle recorded net income of $21.3 million or $1.63 per diluted share and adjusted EBITDA of $125.6 million and non-GAAP earnings per diluted share of $7.54, a significant increase from ...
-
Tris Pharma Reports Positive Topline Data from Clinical Study of Investigational Pain Therapy Cebranopadol Showing Significantly Less Potential for Abuse Versus Tramadol and Oxycodone
Study evaluating human abuse potential demonstrates that the likeability of cebranopadol administered orally in nondependent recreational opioid users was significantly less as compared to immediate release (IR) formulations of either tramadol or oxycodone, suggesting a lower potential for abuse versus traditional C-II and C-IV opioids A first-in-class investigational analgesic uniquely ...
-
Perimeter Medical Imaging AI Announces Publication of Peer-Reviewed Journal Article Validating Potential Use of Wide-Field Optical Coherence Tomography (WF-OCT) in Head and Neck Surgeries
Perimeter Medical Imaging AI, Inc. (TSX-V: PINK)(OTC: PYNKF) (FSE: 4PC) (“Perimeter” or the “Company”) announced the publication of a peer-reviewed research article in JAMA Otolaryngology—Head and Neck Surgery. The study’s findings validate the further exploration of the use of Perimeter’s wide-field Optical Coherence Tomography (WF-OCT) technology to ...
-
gammaCore Sapphire Non-Invasive Vagus Nerve Stimulator (nVNS) Receives Unique National Product Code Number by the Belgian Pharmaceutical Association
electroCore, Inc. (the “Company”), (NASDAQ: ECOR), a commercial-stage bioelectronic medicine company, today announced that gammaCore Sapphire has been issued a National Code Number (CNK) in Belgium. CNK numbers are unique product code identifiers allocated by the Belgian Pharmaceutical Association (APB) for products and medicines commercially available from pharmacies throughout ...
-
ITM Executes Option to In-license Targeted Radionuclide Therapy Candidate LuCaFab (ITM-31) for the Treatment of Malignant Brain Tumors
ITM Isotope Technologies Munich SE (ITM), a leading radiopharmaceutical biotech company, today announced the execution of an exclusive licensing option for LuCaFab (now ITM-31), a novel Targeted Radionuclide Therapy candidate for the treatment of malignant glioblastoma. ITM-31 is a carbonic anhydrase XII (CA XII)-specific antibody Fab fragment developed by Helmholtz Munich and coupled with ITM's ...
-
BioIntelliSense Acquires AlertWatch Clinical Intelligence Engine to Expand its Comprehensive Portfolio of Continuous Patient Monitoring Solutions
BioIntelliSense, a continuous health monitoring and clinical intelligence company, today announced its acquisition of AlertWatch®, an FDA-cleared, clinically proven patient monitoring solution. AlertWatch uniquely provides robust integration, data analysis and a patented intuitive display of contextual patient data for more efficient, actionable clinical decisions. As part of the recently ...
-
100th Patient Enrolled in Clinical Trials for Active Implants’ NUsurface Meniscus Implant
Active Implants, LLC, a company that develops orthopedic implant solutions, today announced enrollment of the 100th patient in clinical trials evaluating the NUsurface® Meniscus Implant for the treatment of persistent knee pain caused by injured or deteriorating meniscus tissue. The surgery was performed by orthopedic surgeon Dr. Wayne Gersoff of Advanced Orthopedics & Sports Medicine ...
-
Active Implants Receives FDA Breakthrough Device Designation for NUsurface Meniscus Implant
Active Implants, LLC, a developer of orthopedic implant solutions for joint preservation, today announced that the NUsurface® Meniscus Implant has been granted a Breakthrough Device Designation from the U.S. Food and Drug Administration. The NUsurface® Meniscus Implant is the first “artificial meniscus” to be marketed in Europe and, if cleared by the FDA, would be the first ...
-
Bexson Biomedical Granted USPTO Patent Allowance on Proprietary Formulation Technology, SeValent
Bexson Biomedical, Inc., a biopharmaceutical company focused on disrupting the small molecule injectable market and enabling home health solutions, today announced the U.S. Patent and Trademark Office (USPTO) has issued a Notice of Allowance for U.S. Application No. 17/546,880 filed on December 9, 2021 and titled Complexing Agent Salt Formulations of Pharmaceutical Compounds. Bexson’s ...
-
Enalare Therapeutics Receives Orphan Drug Designation From the U.S. FDA on ENA-001 for the Treatment of Apnea of Prematurity (AoP)
ENA-001, a New Chemical Entity (NCE) with a novel mechanism of action as an agnostic respiratory stimulant, has previously been granted Rare Pediatric Disease designation for the Treatment of AoP by the FDA, with eligibility for a priority review voucher (PRV). The compound is also being developed by Enalare for the treatment of post-operative respiratory depression and community drug overdose. ...
-
Enalare Therapeutics Receives an Additional Award Worth Up to $50 Million From BARDA, Expanding Its Partnership in Development of ENA-001
The Funding Will Accelerate Development of an Intramuscular Formulation of ENA-001, a Novel Agnostic Respiratory Stimulant, for the Potential Treatment of Community Drug Overdose and as a Medical Countermeasure for Mass Casualty Events. The Funding Supports Development of an Intramuscular ENA-001 From Pre-Clinical Toxicology Through Filing for Approval in the United States Enalare Therapeutics ...
-
Classification of Fractional Lasers
Fractional laser is a commonly used instrument in clinical beauty medicine. In addition to treating acne scars, burn scars, and trauma scars, it also has a good effect on facial rejuvenation, freckle removal, skin shrinking, and stretch marks. Bvlaser is a CO2 fractional laser machine factory supplier, we have best fractional laser machine for sale. What is fractional laser Fractional laser ...
-
Bexson Biomedical Announces Participation in the Jeffries Innovation in Mental Health Summit
Bexson Biomedical, Inc., a biopharmaceutical company focused on disrupting the $189 billion small molecule injectable market and enabling home health solutions, today announced that the Company’s management will be participating in the 2022 Jeffries Innovation inMental Health Summit. Bexson’s Chief Executive Officer, Gregg Peterson, and Chief Scientific Officer, Jeffrey Becker, are ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you