Post-surgery News
-
Showcase
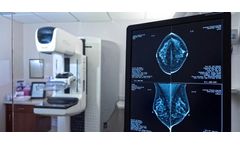
Northern Arizona, Verde Valley MC Highlights VeraForm; Importance Marking Breast Surgical Site
As a patient facing breast cancer surgery, you should know that precise "marking" of your cancer surgical site is an important part of your treatment. A marker is a device inserted at the surgical site. Precision in marking helps make sure you retain as much normal breast tissue as possible with the surgery. This ultimately leads to best overall post-surgical care by your wellness team. A new ...
-
An Easy, Predictable, and Simpler Way to Embolize Tumors
Instylla is now enrolling patients in a clinical trial for the world’s first hydrogel-based liquid embolic intended for the treatment of hypervascular tumors. A second initial application for its Embrace embolic therapy is hemorrhage control, where the product offers advantages in terms of simplicity, speed, and safety. Amar Sawhney and his teams, it seems, can make medical hydrogels do ...
-
7 December 2022 - FDA/CDC highlight need for infection preventions
Particular support for decolonisation of pathogens which is the approach taken by its XF-73 Nasal programme Destiny Pharma plc (AIM: DEST), a clinical stage innovative biotechnology company focused on the development of novel medicines that can prevent life-threatening infections, today reports on the publication of findings from the US Food and Drug Administration (FDA) and US Centers for ...
-
XF-73 Nasal Phase 3 development plans finalised
Targeting peak sales potential of $1 billion in the major markets Destiny Pharma plc (AIM: DEST), a clinical stage innovative biotechnology company focused on the development of novel medicines that can prevent life-threatening infections, is pleased to announce that it has now finalised plans for its Phase 3 studies for its late-stage clinical asset, XF-73 Nasal. Highlights Discussions held ...
-
XF-73 Dermal infection project advances into clinically enabling safety study with US Government’s NIAID
Destiny Pharma plc (AIM: DEST), a clinical stage innovative biotechnology company focused on the development of novel medicines that can prevent life-threatening infections, is pleased to announce the commencement of an Investigational New Drug (IND) enabling safety study with its novel XF-73 Dermal formulation. This study is the second of two planned preclinical safety studies of the XF-73 ...
-
ITM Executes Option to In-license Targeted Radionuclide Therapy Candidate LuCaFab (ITM-31) for the Treatment of Malignant Brain Tumors
ITM Isotope Technologies Munich SE (ITM), a leading radiopharmaceutical biotech company, today announced the execution of an exclusive licensing option for LuCaFab (now ITM-31), a novel Targeted Radionuclide Therapy candidate for the treatment of malignant glioblastoma. ITM-31 is a carbonic anhydrase XII (CA XII)-specific antibody Fab fragment developed by Helmholtz Munich and coupled with ITM's ...
-
Aatru Medical Announces FDA Clearance and Commercial Launch of the NPSIMS - Negative Pressure Surgical Incision Management System
Aatru Medical, LLC ("Aatru") today announced U.S. Food and Drug Administration (FDA) 510(k) Class II clearance of the NPSIMS™ Negative Pressure Surgical Incision Management System. The NPSIMS utilizes an innovative mode-of-action that eliminates the expensive electromechanical pump, battery, and electronics found in most other negative pressure wound therapy (NPWT) systems being deployed ...
-
Empirical Spine Receives FDA Breakthrough Device Designation for the LimiFlex Device Targeting Degenerative Spondylolisthesis
Empirical Spine, Inc., a company developing advanced solutions for the surgical treatment of spinal disorders, today announced that the US Food and Drug Administration (FDA) has granted Breakthrough Device Designation (BDD) for its LimiFlex™ Paraspinous Tension Band. In doing so, the FDA has determined that LimiFlex™ holds the potential to offer significant advantages over ...
-
Ondine completes enrollment for US Phase II nasal photodisinfection trial
Ondine Biomedical Inc. (LON: OBI), has recruited the final patient to its nasal photodisinfection exploratory Phase 2 trial. The trial is evaluating how effectively Ondine’s nasal photodisinfection technology eradicates pathogens – specifically Staphylococcus aureus – in the nose. The single-center, BENEFIT-aPDT open-label study is being carried out at HCA Healthcare’s ...
-
Enalare Therapeutics Receives a National Institutes of Health (NIH) Grant Award
Enalare Therapeutics Inc., a biopharmaceutical company dedicated to developing novel therapies for patients suffering from life-threatening critical care conditions, announced today that it has been awarded a grant from the National Institute on Drug Abuse (NIDA), part of the National Institutes of Health (NIH). Funding from the grant will be used to further the development of ENA001, Enalare's ...
-
Onlume Receives FDA 510(k) Clearance for Fluorescence-guided Surgery Device
OnLume has received 510(k) clearance from the U.S. Food & Drug Administration (“FDA”) to market its first product, a fluorescence guided surgery (FGS) system. The OnLume Imaging System is indicated for fluorescence imaging of blood flow and tissue perfusion before, during, and after vascular, gastrointestinal, organ transplant, plastic, reconstructive, and micro surgeries. This ...
By OnLume Inc.
-
Enalare Therapeutics Announces Positive Topline Results With ENA001 in a Propofol Induced Respiratory Depression Study
Enalare Therapeutics Inc., a biopharmaceutical company dedicated to developing novel therapies for patients suffering from life-threatening critical care conditions, announced today that it has completed a propofol induced respiratory depression study with its lead compound ENA001, having achieved the targeted primary endpoint. The objective of the study was to demonstrate the ability of ENA001 ...
-
Palisade Bio And Newsoara Receive NMPA Clearance To Commence Phase 3 Clinical Trial In China Evaluating LB1148 To Accelerate The Return Of Bowel Function Following Abdominal Surgery
NMPA clearance expands global Phase 3 clinical development program; Company recently announced clearance from U.S. Food and Drug Administration to commence Phase 3 study Newsoara to fully fund Phase 3 program in China Under the Co-Development Agreement, commencement of Phase 3 program in China will trigger milestone payment to Palisade Bio Palisade Bio (Nasdaq: PALI), a clinical stage ...
By Palisade Bio
-
Palisade Bio (Nasdaq: PALI) To Participate In The Virtual Investor Management And KOL Roundtable Event
- Palisade Bio (Nasdaq: PALI), a clinical stage biopharmaceutical company advancing therapies for acute and chronic gastrointestinal (GI) complications, today announced that members of management will participate in the Virtual Investor Management and KOL Roundtable Event on Tuesday, May 3, 2022 at 10:00 AM ET. Participating from the Palisade Bio management team will be Tom Hallam, Ph.D., Chief ...
By Palisade Bio
-
Biostage Further Strengthens IP Portfolio with Additional Patent for Regeneration and Repair of the Bronchus
Biostage, Inc. (OTCQB: BSTG) ("Biostage" or the "Company"), a cell-therapy biotechnology company with successful first-in-human experience in treating esophageal cancer (conducted at the Mayo Clinic and published last August) and FDA approval to commence a clinical trial of the Biostage Esophageal Implant for severe esophageal disease including cancer, today announced that the U.S. Patent and ...
-
Sight Sciences Reports Fourth Quarter and Full Year 2021 Financial Resultsgrafic lines 4 Sight Sciences
Sight Sciences, Inc. (Nasdaq: SGHT), an eyecare technology company focused on creating innovative solutions intended to transform standards of care and improve patients’ lives, today reported financial results for the quarter and year ended December 31, 2021. Recent Business Highlights Generated 2021 total revenue of $49.0 million, an increase of 77% compared to the prior year ...
-
Palisade Bio (Nasdaq: PALI) Announces Analysis Demonstrating LB1148 Reduced The Extent And Severity Of Post-Surgical Intraabdominal Adhesions By 93%
Data from Pooled-Analysis of Enteral Protease Inhibitor LB1148 Demonstrated 72% Reduction in the Risk of Adhesion Formation in Patients Undergoing Bowel Resection Surgery Company to Host Conference Call on Wednesday, March 16, 2022, at 8:30 a.m. EST Palisade Bio, Inc. (Nasdaq: PALI), a clinical stage biopharmaceutical company advancing therapies for acute and chronic gastrointestinal (GI) ...
By Palisade Bio
-
A Brand New Collaboration Between Emovi and The Geneva Foundation
In partnership with The Geneva Foundation and supporting grant from the Quebec Ministry of Economy and Innovation, Emovi will launch the 24-month clinical project within US Army clinics at Fort Bragg and High Point University, North Carolina, USA Emovi, Inc., a leading medical device developer and manufacturer for orthopaedics, announced today a clinical collaboration with the US Army in ...
By Emovi
-
Palisade Bio (Nasdaq: PALI) To Present New Clinical Data Of LB1148 For Preventing Adhesions In Conjunction With Next Big Thing Session At Society Of American Gastrointestinal And Endoscopic Surgeons 2022 Annual Meeting
Palisade Bio, Inc. (Nasdaq: PALI), a clinical stage biopharmaceutical company advancing therapies for acute and chronic gastrointestinal (GI) complications, was selected to present an ePoster entitled “Pooled-Analysis of Incidence and Severity of Post-Surgical Intraabdominal Adhesions following Bowel Resection Surgery and Treatment with Enteral Protease Inhibitor LB1148” at The Next ...
By Palisade Bio
-
Healthium in collaboration with Abhinav Bindra Foundation Trust (ABFT) inaugurates the ‘Sport of Life’, Bengaluru
The “Sport of Life” initiative aims to promote awareness about sports injuries and provide treatment for sportspeople. As a part of this initiative, Healthium will aid in the treatment of sports injuries with free arthroscopy implants. Bhaichung Bhutia, torchbearer of Indian Football in the international arena, Abhinav Bindra, India's first Individual Olympic gold medalist and ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you