

Rocket Pharmaceuticals, Inc.
- Home
- Companies
- Rocket Pharmaceuticals, Inc.
- Products
- Adeno-Associated Viral Vector (AAV) ...
Adeno-Associated Viral Vector (AAV) Therapy
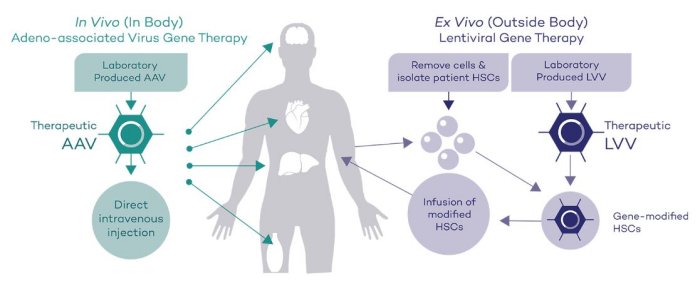
The LVV platform is ideal for modifying hematopoietic stem cells (HSCs) to address disorders affecting the bone marrow. The LVV transduction process occurs ex vivo (outside the body), which ensures that the gene has been properly integrated before the therapy is given to the patient. The process involves collection and isolation of a patient’s hematopoietic stem cells (HSCs), insertion of the corrected gene into the HSCs via a lentiviral vector outside the body, and the infusion of modified HSCs back into the patient. The goal is to enable sufficient quantities of a healthy, disease-modifying therapeutic protein to be manufactured by the patients’ own hematopoietic cells. Rocket’s LVV-based programs include: Fanconi Anemia, Leukocyte Adhesion Deficiency-I, Pyruvate Kinase Deficiency and Infantile Malignant Osteopetrosis.
Most popular related searches
adeno-associated virus
hematopoietic cells
hematopoietic stem
hematopoietic cell
stem cell
clinical stage
bone marrow
lentiviral vector
viral vector
anemia
Discovery
Rocket’s Research and Discovery group in the Cranbury, New Jersey facility utilizes approximately 20,000 square feet of state-of-the-art laboratories for designing, optimizing and advancing candidate therapeutics to clinical stage development.
Manufacturing
Rocket’s manufacturing facility in Cranbury, New Jersey was designed specifically for Rocket’s manufacturing needs. Approximately half of the newly constructed 103,720 square foot facility will be dedicated to adeno-associated virus (AAV) Current Good Manufacturing Practice (cGMP) manufacturing.
