

- Home
- Companies
- Indee Labs
- Products
- Hydropore - Non-Viral Technology

Hydropore - Non-Viral Technology
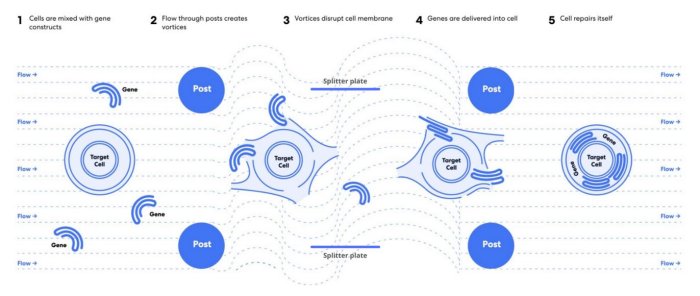
Hydropore™ is a simple, scalable and efficient way to accelerate the discovery, development and manufacturing of gene-modified cell therapies (GMCTs) such as T cell immunotherapies. This novel, non-viral technology utilizes the power of microfluidic vortex shedding (µVS) to rapidly deliver nucleic acids, proteins and gene-editing complexes to immune cells in a matter of seconds, yielding millions of high quality cells with negligible impact to cell viability and function. Hydropore™ bypasses the challenges and limitations of viral transduction and traditional transfection via electroporation, allowing companies to accelerate their research goals from discovery to manufacturing.
The future of engineered cell therapies starts here.
Power your research with our patented, non-viral technology to rapidly deliver nucleic acids, proteins and gene-editing complexes to immune cells while yielding millions to billions of high quality cells with preserved cell viability and function.
Order Hydropore™ for knockout and knock-in genome editing now!
Hydropore™ is developed in a regulatory-ready manner and currently available for Research Use Only. A clinical system will be regulated as a tool for biologics manufacturing. Both systems are developed in an ISO 13485-compliant manner and all materials in the flow path are medical-grade or USP Class VI implantable-grade. Hydropore™ is also suitable for high-throughput pooled screening of small volumes.
Power your research with microfluidics.
Order Hydropore™ for knockout and knock-in genome editing and join 3 top pharmaceutical companies using the Hydropore™ platform.
Hydropore™ is a non-viral, platform technology using hydrodynamic conditions termed microfluidic vortex shedding (μVS) to gently permeabilize cells, allowing for quick and efficient delivery of payloads such as DNA, mRNA and CRISPR Cas9-RNPs into immune cells for gene-editing applications. Our patented technology is a robust alternative to electroporation and viral transduction that addresses current industry challenges in cell therapy manufacturing by relieving the bottleneck of the gene transfer step. Companies can circumvent high development costs, long lead times, and increased risk & regulation while still yielding millions of high quality, engineered cells with minimal impact to cell viability and function.
- Preserved Cell Viability & Function — High yield of recovered, viable & modified cells with minimal impact to the native cell state.
- High Speed & Throughput Capability — Each 5 x 10mm chip can process >50 million cells at >2-4 million cells per second and as low as 100uL in processing volume.
- Simple and Scalable — Easy-to-integrate with existing research, clinical & commercial workflows. μVS devices can be readily manufactured at scales of >2k per day.
- Strong Clinical Applicability — Devices are developed in an ISO 13485-compliant manner and all materials in the flow path are medical-grade or USP Class VI implantable-grade.
