Arterial Vessel (Arterial Health) Equipment
-
Manufactured by Mizuho Medical Co., Ltdbased in JAPAN
New High Sensitive Fluorescence camera displays Fluorescence and Visible Light Images Simultaneously. ...
-
Manufactured by Cerevast Medical, Inc.based in USA
When a blood clot or thrombus is formed, strands of fibrin anchor one red blood cell to another. It is these fibrin strands that form the lattice or structural matrix of the blood clot. The formation or deposition of these clots in the arterial vessels of the brain is one of the primary causes of ischemic stroke. Intravenous thrombolytic ...
-
Manufactured by Biosensors International Group, Ltd.based in SINGAPORE
The use of the BioStream PTCA catheter is indicated for patients with discrete lesions of the coronary arteries with a reference vessel diameter of 1.50 to 4.00 mm for the purpose of improving the coronary lumen diameter. The BioStream PTCA catheter is indicated for the dilatation of the affected segments of a coronary artery or a coronary ...
-
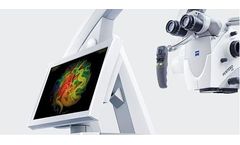
Manufactured by Carl Zeiss Meditec AGbased in GERMANY
feeding arteries, nidal vessels and draining veins – providing objective access to speed and sequence of blood ...
-
Manufactured by SCIREQ - an emka TECHNOLOGIES Companybased in CANADA
emkaBATH is a type of tissue bath setup, designed for contractile measures in tissues such as aortic, tracheal, iliac, and papillary muscles, and/or microvessels such as veins, small arteries, and lymph vessels. The system provides heated, oxygenated, and nutrient-rich perfusate to the tissue with or without drug administration and ...
-
Manufactured by Inspital Medical Technology GmbHbased in GERMANY
is 96 which is the perfect rate. Having a CRI index close to 100 is important for the surgeon in order to better recognize the details in the wound area, to recognize tissue colors of arterial and venous vessels more easily. Thanks to its special lamp head design, it has an aerodynamic structure working with positive pressure bacterial ...
-
Manufactured by HAGMEDbased in POLAND
Catheters are used to remove fresh, soft embolic material from blood vessels in the arterial system, administer thrombolytic or anticoagulant drugs, and administer contrasting fluids into arter – ies, occlusion of blood vessels in the arterial system, and blood aspiration from the blood vessels of the ...
-
Manufactured by Amsel Medical Corporationbased in USA
CE mark and FDA cleared SCureClamp with Interdigitation Occlusion Technology (iDOT) is designed to easily and securely close vessels – veins, arteries, and tubular structures – during surgical and interventional procedures. This unique mechanical surgical clamping technology is delivered directly through a fine, minimally ...
-
Manufactured by Starr Life Sciences Corp.based in USA
The STARR-Link Analog Output Module provides 0-5 Volt analog outputs of the parameters measured by the MouseOx. It serves as an interface between the MouseOx and other data acquisition hardware. Output of MouseOx Data, Voltage Range of 0 to 5, Up to 8 Simultaneous Data Channels, No Calibration Required. The?STARR-Link?is an?Analog Data Output Module?for the?MouseOx Plus?that?converts the ...
-
Manufactured by Elixir Medical Corporationbased in USA
The DynamX Drug Eluting Coronary Bioadaptor System is a significant innovation in the treatment of coronary artery disease. Going beyond drug-eluting stents (DES), DynamX represents one of the most significant breakthroughs in implant design in the past 30 ...
-
Manufactured by P+F Products + Features GMBHbased in AUSTRIA
in the coronary artery by inflating the balloon. It is intended as a permanently implanted, non-reusable device. ...
-
Manufactured by Caladrius Biosciences, Inc.based in USA
Also known as coronary microvascular disease, microvascular angina, cardiac syndrome x, ischemic heart disease, non-obstructive coronary artery disease, small vessel disease, small artery disease, and ischemia with no obstructive coronary arteries (INOCA) Coronary microvascular dysfunction (CMD) is a disease that causes narrowing ...
-
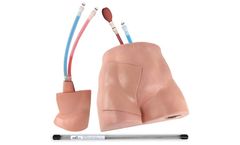
Manufactured by Simulab Corporationbased in USA
Simulab's FemoraLineMan System is an ultrasound-compatible femoral line vascular access trainer. The product offers an effective training solution for central venous or arterial access at the femoral site. ...
-
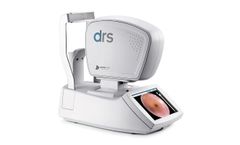
Manufactured by iMEDOS Health GmbHbased in GERMANY
The systems of static vessel analysis offer an innovative solution for the evaluation of the condition of vessels. Designed for use in clinical routine, they provide important parameters and information on patients’ individual risk of developing cardiovascular diseases and events (e.g. heart attack and stroke), diabetes mellitus and ...
-
based in USA
The Alucent NVS Vessel Restoration System is designed to create a natural, stent-like scaffold using the body’s own tissue and processes. An all-in-one, integrated procedural system, Alucent NVS combines standard angioplasty techniques with a small molecule drug that when photoactivated, promotes extracellular matrix protein linking ...
-
Manufactured by Simulab Corporationbased in USA
Simulab's FemoraLineMan Training Package is an ultrasound-compatible task trainer that offers an effective training solution for central venous or arterial access using the femoral site. This femoral line trainer uses the same patented technology as the highly acclaimed TraumaMan System and allows medical professionals to train using ...
-
Manufactured by Veryan Medical Ltd.based in UNITED KINGDOM
The Challenge: Peripheral Arterial Disease. People are living longer and with an increase in the incidence of obesity, diabetes and other contributing factors, claudication may cause substantial tissue changes and, in some scenarios, can lead to amputation and even death. The peripheral stent market is forecast to grow to $2 billion by 2024 ...
-
Manufactured by Abbott Laboratoriesbased in USA
Accuracy where it counts. Exact placement, strength and performance where it counts. Stability; Multiple points of contact between filter and vessel for optimal wall apposition Filter wire torqued independently from filter Optimal protection Homogenous laser-cut holes Large capture volume while allowing blood flow Visibility Four ...
-
Manufactured by Carl Zeiss Meditec AGbased in GERMANY
INFRARED 800 from ZEISS enables intraoperative visual assessment of blood flow and vessel patency during arteriovenous malformation (AVM), bypass, aneurysm and anastomosis surgery. ZEISS INFRARED 800 is indicated for use in neurosurgery, plastic and reconstructive procedures and coronary artery bypass graft ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you