Microvascular News
-
Showcase
VisionQuest Abstract on Diabetic Peripheral Neuropathy Accepted by ADA
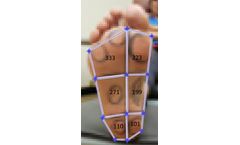
We’re looking forward to presenting “Detecting Diabetic Peripheral Neuropathy Utilizing Thermoregulation of the Plantar Foot” at the upcoming 82nd Scientific Sessions of the American Diabetes Association. Our research aims to detect thermal (infrared) biomarkers of diabetic peripheral neuropathy on the feet of diabetic subjects who have not been diagnosed with DPN. By analyzing ...
-
Gesynta Pharma completes patient enrollment in its Phase II study in systemic sclerosis
Gesynta Pharma AB today announces that all patients have been recruited for the company's clinical Phase II study with the drug candidate GS-248, which is being evaluated as a treatment for the rare disorder systemic sclerosis.The study is intended to demonstrate proof-of-concept and to evaluate the safety profile of GS-248. Top-line data is expected in the fall of 2022. Systemic ...
-
Caladrius Biosciences Provides Update on Phase 2b FREEDOM Trial of XOWNA® in Coronary Microvascular Dysfunction
BASKING RIDGE, N.J., May 23, 2022 (GLOBE NEWSWIRE) -- Caladrius Biosciences, Inc. (Nasdaq: CLBS) (“Caladrius” or the “Company”), a clinical-stage biopharmaceutical company dedicated to the development of innovative therapies designed to treat or reverse disease, today announced that the Company has suspended patient enrollment in its Phase 2b study of XOWNA®, known as ...
-
Gesynta Pharma`s drug candidate GS-248 granted Orphan Drug Designation in US by the FDA for the treatment of systemic sclerosis
Gesynta Pharma AB today announces that the U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation to the company's drug candidate GS-248 for the treatment of systemic sclerosis. GS-248 is currently being evaluated in a Phase II clinical trial as a treatment for Raynaud's phenomenon secondary to systemic sclerosis. Orphan Drug Designation is intended to encourage the ...
-
Miracor Medical starts 2nd randomized study, targeting expanded indications
Miracor Medical SA (Miracor Medical) today announced the first patient enrolled in the PICSO-AMI-V study to evaluate the benefits of PiCSO® (Pressure-controlled intermittent Coronary Sinus Occlusion) therapy as an adjunct to conventional primary percutaneous coronary intervention (PCI) for patients presenting with inferior ST-Elevation Myocardial Infarction (STEMI). The first patient ...
-
FDA clears Sonavex’s ultrasound blood flow monitor
Sonavex scored FDA clearance for its technology that uses ultrasound imaging and deep learning to deliver blood flow data on demand. The EchoSure system is cleared for use with the company’s EchoMark bioresorbable tissue markers, which got the agency’s nod in June. “The key goal is to be able to detect the problems associated with microvascular and vascular surgeries with ...
-
Hemostemix to combine ACP-01 with Dr. James Shapiro’s Islet Cells to treat Type 1 Diabetes
Hemostemix Inc. (“Hemostemix” or the “Company”) (TSXV:HEM; OTCQB:HMTXF; FSE:2VFO) is pleased to announce it has signed a contract with Dr. James Shapiro, University of Alberta, Edmonton and will complete a transfer ACP-01 to Dr. Shapiro’s laboratory. The combination of ACP-01, an autologous angiogenic cell precursor that has demonstrated improvement of angiogenesis ...
-
Phase II update: Gesynta Pharma announces significant progress across development program for first-in-class drug candidate GS-248
Gesynta Pharma AB today announced that an IND (Investigational New Drug Application) has been submitted to the U.S. Food & Drug Administration for its oral drug candidate GS-248 in patients with systemic sclerosis. Concurrently, more than half of the patients in the ongoing Phase II study in four countries across Europe have been recruited. The study investigates the safety of GS-248 and ...
-
Gesynta Pharma initiates Phase II study of its first-in-class drug candidate GS-248 in patients with systemic sclerosis
Gesynta Pharma AB today announced that the first patients have been dosed in a Phase II study of its oral drug candidate GS-248 in patients with systemic sclerosis - a debilitating autoimmune disease that causes serious damage to the microvasculature. This proof-of-concept study will investigate the safety of GS-248 and its efficacy on Raynaud's phenomenon and peripheral blood flow in this ...
-
MagicTouch SCB Granted ‘Breakthrough Device Designation’ for the treatment of Small Coronary Artery Lesions.
Concept Medical Inc. (CMI) has been granted “Breakthrough Device Designation” from the Center for Devices and Radiological Health (CDRH) of the Food and Drug Administration (FDA) for the MagicTouch SCB Sirolimus Coated Balloon Catheter, for the treatment of small coronary artery lesions in Coronary Artery Disease (CAD). The proposed indication for use includes “The ...
-
Miracor Medical announces PiCSO presentations during PCR e-Course
Miracor Medical SA announces today that an update about PiCSO therapy will be presented during the upcoming PCR e-Course which will be held on June 25, 26 and 27. PiCSO therapy (Pressure-Controlled Intermittent Coronary Sinus Occlusion) is delivered by interventional cardiologists during the primary PCI (Percutaneous Coronary Intervention) procedure in patients experiencing anterior ST-elevated ...
-
Miracor Medical awarded CE Mark for PiCSO Impulse Catheter and Console
Miracor Medical SA (Miracor Medical) has announced the award of the CE Mark for its latest generation of PiCSO Impulse Catheter and PiCSO Impulse Console, which is indicated for the treatment of anterior STEMI patients. The new system features improvements in ease of use. PiCSO therapy is delivered by interventional cardiologists during the primary PCI (Percutaneous Coronary Intervention) ...
-
Gesynta Pharma reports positive Phase I results with lead candidate GS-248 for microvascular disease at EULAR 2020
Gesynta Pharma AB ("Gesynta"), a clinical stage company developing novel anti-inflammatory agents, today announced that the results from the successfully completed First-in-Human clinical study with its lead candidate GS-248 for the treatment of microvascular disease have been presented at the EULAR 2020 E-Congress. Following these results, Gesynta now intends to commence a Phase II study in ...
-
Gesynta Pharma announces positive Phase I results with GS-248 for the treatment of microvascular disease - data to be presented at EULAR congress
Gesynta Pharma AB ("Gesynta") announces the completion and positive results of a first clinical study with GS-248. The purpose of the study was to evaluate safety, tolerability, pharmacokinetic and pharmacodynamic properties of GS-248 after single and multiple ascending doses to healthy subjects. The results support the further clinical development of GS-248 in microvascular dysfunction and a ...
-
Research Presented at LINC Suggests the Profusa Lumee® Oxygen Platform May Improve Clinical Management of Patients with Critical Limb Ischemia
Profusa, a digital health company that is pioneering the next generation of personalized medicine, today announced research findings that suggest the company’s Lumee® Oxygen Platform may help improve the clinical management of patients with critical limb ischemia (CLI) who are undergoing endovascular revascularization treatment (EVT). The data, from a recent post-market clinical study ...
-
Mauna Kea Technologies Receives FDA 510(k) Clearance of Cellvizio® with a Fluorescent Dye, Fluorescein, as Drug Device Combination
The Cellvizio®100 series system with all its different Confocal Miniprobes™ is now cleared for use with the fluorescein dye to image blood flow in the microvasculature and capillaries Mauna Kea Technologies today announced that it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for the Cellvizio® 100 series and all associated Confocal Miniprobes™ for ...
-
Miracor Medical starts PiCSO-AMI-I randomized study in EU
Miracor Medical SA (Miracor Medical) today announced the initiation and first patient enrollment in the PICSO-AMI-I study to evaluate the benefits of PiCSO® therapy compared with conventional PCI for the treatment of anterior STEMI patients. The first patient enrolled into this study was treated at Oxford Heart Center, Oxford, UK by Professor Adrian Banning, the principal investigator of the ...
-
Gesynta Pharma initiates clinical phase I study with GS-248 for the treatment of microvascular disease
Gesynta Pharma AB ("Gesynta") announces that the first clinical study with GS-248 has been initiated and that the first healthy volunteers have been dosed. The purpose of the phase I study is to study safety, tolerability and pharmacokinetic properties of GS-248 as well as its effect on relevant biomarkers. Gesynta's lead candidate drug, GS-248, is a potent and selective inhibitor of ...
-
Sonavex Receives FDA 510(k) Clearance for EchoSure
Sonavex, Inc., a privately held medical device company focused on empowered patient care, announced today that on March 8, 2019 it received 510(k) clearance from the U.S. Food and Drug Administration for its EchoSureTM device to deliver definitive blood flow data on demand. The EchoSure system combines 3D ultrasound imaging with advanced deep learning algorithms to automate visual and ...
-
Miracor Medical closes final tranche of Series-D bringing the round to 30€M
Miracor Medical SA (Miracor Medical) today announced the final closing of the Series D financing round of €30M. The Series D capital will be used to further develop and commercialize the PiCSO® Impulse System. The Series-D round was led by Ming Capital (Shenzhen, China) and co-led by a strategic investor. Participating new investors include Belgian alternative investment fund Quest for ...
Need help finding the right suppliers? Try XPRT Sourcing. Let the XPRTs do the work for you